Overview
Antibiotics and antibiotic resistance
Antibiotics are medicines that have activity against bacteria. They may function by either killing the bacteria (bactericidal) or by inhibiting the growth and proliferation of bacteria (bacteriostatic) allowing the animal’s immune system to more effectively fight a bacterial infection. In either case, the bottom line is that an antibiotic treatment stops the growth of a bacterial infection so the host (i.e. the animal) can eliminate it. The animal can then recover and return to health.
LEARN MORE
There are several ‘classes’ of antibiotics. Penicillins, tetracyclines and macrolides are some common classes of antibiotics. Some are effective against a wide range of bacteria (broad spectrum), while others may target only a small set of bacteria (narrow spectrum). Some types can be used in both people and animals, while some are only useful and approved in animals or people. The term ‘medically important’ can refer to antibiotic classes important to human medicine. This categorization of importance has been defined by the FDA. A subset of these antibiotics is also important to animal medicine. Many antibiotics important to human medicine are not used in animals. Additionally, there are other antibiotic classes used in animal medicine (e.g., ionophores) that are not used, nor are they important to human medicine. A separate listing of antibiotic classes important to animal medicine has been defined by the OIE.
An antibiotic will kill or inhibit the growth of bacteria that are susceptible to it. Antimicrobial resistance (AMR) to antibiotics occurs when the disease-causing bacteria change in some way to reduce or eliminate the effectiveness of the antibiotic used to treat infections. Sometimes bacteria have natural, or inherent, properties that make them resistant to an antibiotic. The concern is for those bacteria that are inherently susceptible to an antibiotic but then acquire genes that enable them to withstand the effects (be resistant to) that same antibiotic. Bacteria can acquire a resistance to one or more types of antibiotics but still be susceptible to other types of antibiotics.
The public health concern occurs when an animal or human illness is caused by bacteria that are resistant to so many antibiotics that the veterinarian or doctor cannot treat the patient with an antibiotic that will be effective.
Antibiotics are so important in treating many common and serious diseases that antibiotic resistance is considered one of the biggest global health emergencies. If it is not properly managed, it could put commonly used antibiotics at risk and turn minor, treatable infections into major, lasting health threats. There are also fears that bacterial infections will acquire multiple resistance mechanisms, leaving no antibiotic treatment options that can be used successfully for some patients.
AMR can be a threat to both animals and people. Many bacterial diseases are zoonotic, meaning that these diseases can be passed back and forth between people and animals. As a result, strategies to contain AMR need a “One Health” approach, meaning physicians and veterinarians must work together to address the challenge of antibiotic resistance.
Responsible antibiotic use is a means to manage AMR and preserve these medicines for the future. It is important to always practice the best possible antimicrobial stewardship, and the animal health industry is committed to providing veterinarians with the most current information about antibiotic products to allow them to make the appropriate stewardship decisions.
Antibiotic Use in Animals and People
Why and how are antibiotics used
Animals are vulnerable to some of the same bacterial infections as people, such as pneumonia and skin infections, and can be treated with antibiotics. Just like in people, antibiotics are used in animals to treat, control and prevent diseases.
LEARN MORE
Many diseases spread by ticks can affect both animals and people. Antibiotics are often given to treat these infections, which can be chronic and cause severe illness.
There are life stages when the risk of bacterial infection is higher. Like in people, a young animal is more likely to succumb to an illness because their immune system is still maturing. Likewise, times of stress lower the body’s defenses and an infection is more likely to occur. Examples of times of stress in an animal’s life include transportation or change in location, rapid change or extreme weather or following surgery. For this reason, farm animals may more likely require antibiotics at certain times in their lives whether to treat an existing infection or to control the spread of a bacterial disease within a group of animals. Farmers and veterinarians work together to prevent these occurrences and create and maintain healthy conditions for their flocks and herds. They are constantly working to improve biosecurity, vaccinate and adopt other management techniques to maintain a healthy herd and keep their animals from getting sick. Despite these efforts, disease threats still occur, and veterinarians need access to antibiotics to address illnesses and protect the welfare of the individual and the herd.
How an antibiotic is given to a patient, whether a person or an animal, is called the “route of administration.” Antibiotics for animals can be administered in the same way as for people: orally or via an injection.
Administering antibiotics to animals can be challenging. While a person can usually take individual responsibility for their medicine, animals must be treated by a person. Anyone with a pet knows that administering a medicine to an animal can be a challenge. And there are practical considerations when administering antibiotics to large groups of farm animals.
When there is a disease affecting a large group of animals, veterinarians must use group treatment, meaning administering antibiotics through either feed or water to be sure the animals are receiving the treatment. In this case, the veterinarian uses information about the farm, size, and weight of the animals, and feeding regime to recommend the amount of medicated feed or water to provide to ensure a proper dosage. Then, to appropriately administer an antibiotic a farmer must ensure his animals get the right amount of dose at the right time and frequency without causing the animal unnecessary stress.
Approval and Regulation of Animal Antibiotics
Like antibiotics for humans, antibiotics for animals are heavily regulated
The FDA reviews and approves animal medicines using the same standards and processes as those used for human medicine.
LEARN MORE
Companies seeking approval of antibiotics for animals submit data to FDA demonstrating that the product meets three standards:
- Ensure the product is safe. Safety, according to FDA, includes safety to the animals, safety of food products derived from the animals, safety to persons administering the drug or otherwise associated with the animals, and safety in terms of the drugs impact on the environment.
- Ensure the product is efficacious. A product sponsor provides data for FDA review and concurrence to demonstrate that the product is safe for the proposed claim or indication.
- Ensure that a quality product is manufactured. Quality means the company adheres to the Good Manufacturing Principles and demonstrates an ability to consistently provide a stable product that is uniform in potency.
These requirements apply to all products approved by FDA. Antibiotics require an additional step, which is an evaluation of how use of the antibiotic might impact the prevalence of AMR.
Government oversight does not end with FDA approval. Three additional components to government monitoring of antibiotics used in food animals are listed below:
- Companies must report to the FDA any developments in what is known about the product, including new studies that are published, new data that comes to light or any problematic reactions that develop in treated animals.
- The U.S. Department of Agriculture monitors meat for the occurrence of antibiotic residues. This monitoring program acts as a check to ensure antibiotics are being used properly and to ensure the FDA-mandated withdrawal times are followed.
- The FDA operates the National Antimicrobial Resistance Monitoring System (NARMS) which tracks antibiotic resistant pathogens in samples collected from animals at slaughter, retail meats and people. The findings, published annually, provide information to policymakers on potential emerging resistance.
The FDA approves all claims (what it is used for) and directions (how it is used) that are on the product label. FDA approves antibiotics with one of three claims:
- Disease treatment – the administration of antibiotics to sick animals to resolve disease in the individual and(or) treatment of outbreaks within the entire herd or flock.
- Disease control – administration to groups of animals to contain the spread of a disease when some animals in the flock or herd become ill and expose the rest of the animals to a bacterial pathogen.
- Disease prevention – administration when animals are not sick but there is sufficient history or risk of disease to warrant therapy under the direction of a veterinarian.
Since the label also lists a specific disease or pathogen against which the antibiotic works, FDA has stated they consider each of these claims to be prudent and judicious therapeutic claims for the use of antibiotics in food producing animals.
The label also contains information on the proper use of the product, such as the disease target, the dose needed for the specific animal according to its weight, and the route of administration. The label may also contain restrictions or constraints on use, such as the withdrawal time. For example, the label will include withdrawal time, or period of time during which antibiotics may not be administered before the animal or animal products can enter the food supply. The withdrawal time is mandated to ensure the meat or milk does not contain residues above limits set by FDA.
Veterinarians and Animal Antibiotics
All medically-important antibiotics used in animal feed and water are under the control of licensed veterinarians.
Licensed veterinarians play an important role in antibiotic management.
Dr. Clara Nelson; addresses food supply and disease prevention via vaccines (2016)
LEARN MORE
Medically important antibiotics used in feed and water for food animals all have two significant restrictions: First, they can only be used under the direction of a licensed veterinarian using a Veterinary Feed Directive (VFD). A VFD is a form filled out by a veterinarian who has determined an antibiotic should be administered through an animal’s feed. The VFD gives directions to a feed mill to mix antibiotics with feed, at the proper dose, and allows it to be administered to the animals. Second, antibiotics used in this manner may only be used according to the FDA-approved directions on the label. The veterinarian cannot use the product in an off-label manner, meaning the dosage and use directions on the label must be followed exactly.
Reducing the Risk to Humans
Interventions reduce the risk of bacteria transferring from food animals to people
Proven policies and procedures help reduce the possibility of bacteria transferring to people via the food supply.
LEARN MORE
Bacteria – both resistant and susceptible – can pass between humans and animals. The concern about the use of antibiotics in food animals is that this use will create selection pressure leading to resistant bacteria in the animals that can be transferred to humans via consumption of food products from those animals.
There are many policies and procedures in place to reduce the possibility of bacteria transferring to people via the food supply. These include:
- The stringent drug approval process at FDA,
- USDA’s Hazard Analysis/Critical Control Points (HACCP) procedures to remove pathogens in processing plants;
- The USDA residue monitoring program; and,
- Good cooking and food handling instructions
Agriculture Doesn’t Pose the Greatest Risk to Humans
Risk of antibiotic resistance is mostly due to human healthcare use
According to the U.S. Centers for Disease Control (CDC), overuse and misuse of antibiotics in all settings allows the development of resistant bacteria. While all uses of antibiotics can exert selection pressure for resistant organisms, research shows the risk of antibiotic use in animals is extremely low compared to uses in human healthcare settings.
LEARN MORE
In 2013 the U.S. Centers for Disease Control published a report titled “Antibiotic Resistance Threats.” This document listed the most urgent bacterial resistance threats faced by doctors in human healthcare settings. Most are pathogens that don’t occur in animals and thus using antibiotics in animals does not affect the prevalence of these. A couple examples:
- One of the bacterial threats listed as “urgent” is Clostridium difficile (C. difficile) which causes life-threatening diarrhea. These infections mostly occur in people who have had both recent medical care and antibiotics. Often, C. difficile infections occur in hospitalized or recently hospitalized patients. This bacterium does not occur in animals.
- Streptococcus pneumoniae (S. pneumoniae, or pneumococcus) is the leading cause of bacterial pneumonia and meningitis in the United States. It also is a major cause of bloodstream infections and ear and sinus infections.
These are examples of serious antibiotic resistance threats that cannot be associated with animals.
In the CDC report only 2 of the 18 antibiotic resistant threats– Campylobacter and Salmonella are associated with farm animals. These are bacteria that commonly reside in the gut of animals and can make people sick if they are consumed in raw or undercooked meat. Because these two bacteria can reside in animals, they are the focus of attention and interventions by the animal health industry, producers and veterinarians. Two studies published in Critical Reviews in Science and Nutrition focus on what is published in scientific literature about these two pathogens:
- In 2016 a review study on Campylobacter aligned with NARMS reports, finding “contamination on most retail meats was exceedingly low.” The research team found no conclusive evidence of a definitive link between use of antibiotics in food animals and emergence of drug-resistance Campylobacter. Campylobacter infections have been linked directly to drinking raw milk or easting food products made from raw milk.
- In 2017 a similar review was published on Salmonella, finding the overall prevalence of drug-resistant? Salmonella summarized in the systematic review aligned with recent National Antimicrobial Resistance Monitoring System (NARMS) reports. The 2013 NARMS report showed that 81% of the Salmonella from human infections carried no resistance to any antibiotic, while Salmonella resistance rates in animals varied by the antibiotic tested. The findings of this systematic review did lead the authors to cite important concerns about Salmonella and call for more research in this area For example, six articles showed increased antibiotic resistance in organisms derived from animals, not retail meats, used in conventional farming, versus those from antibiotic-free operations. No studies were found that followed animal-associated antibiotic resistant isolates from farm to retail products.
Antibiotic resistance is a broad public health concern with many facets and cannot be adequately addressed by focusing only on animal use. Research on measures to restrict use in animals in Denmark shows interventions have not decreased resistance in key zoonotic bacteria. Other research shows that if action is only taken in animals, AMR in people will likely remain unchecked.
More research is needed to fully understand the relationship between drug resistant bacteria in people and animals. However, until then, we must prioritize responsible use and actions with measurable results in both animals and people.
Animal Health Industry Action on Antibiotic Resistance
The animal health industry is taking action to ensure responsible antibiotic use in animals to help limit resistance
The animal health industry works with veterinarians and producers to ensure judicious use of antibiotics in food animals.
LEARN MORE
The animal health industry is dedicated to working with veterinarians and producers to ensure judicious use of antibiotics in food animals.
Animal health companies and our trade associations worked collaboratively with the FDA to implement the agency’s Judicious Use Policy in 2017. Under this policy, companies voluntarily removed growth promotion claims from medically important antibiotic compounds, leaving only therapeutic claims. As a result, antibacterial agents medically important to humans are used in food animals only to fight disease.
Also, as part of Judicious Use policy, all the antibiotics products with therapeutic claims that are used in feed and water were brought under the supervision of a licensed veterinarian. All medically important antibiotics added to feed must be approved by a veterinarian by providing a Veterinary Feed Directive (VFD), written instructions to a feed mill on how to properly mix antibiotics in the feed. The veterinarian must have a Veterinary Client-Patient Relationship (VCPR) with the producer of the animals and must make the medical judgment that administration of the antibiotic is needed due to disease threats or outbreaks.
In 2017, the global animal health industry released our “Commitments and Actions on Antibiotics Use.” It outlines our five guiding principles for responsibly using antibiotics and tackling antimicrobial resistance.
We continue to demonstrate our commitment by working with the FDA on the agency’s Five-Year Plan for Supporting Antimicrobial Stewardship in Veterinary Settings. This plan includes, among other things, bringing all remaining antibiotics that are marketed over-the-counter under prescription status, thus ensuring veterinary oversight; ensuring labels on medically important compounds have appropriate duration of use instructions; collecting and analyzing data on the use of antibiotics in animals; and the development of a strategy to ensure antimicrobial stewardship in companion animals.
Reducing the Need for Antibiotics
The animal health industry is committed to researching new products and ways to reduce the need for antibiotics
The animal health industry is committed to seeking new ways to reduce the need for antibiotics.
LEARN MORE
The animal health industry is committed to working with veterinarians and producers to protect the health and welfare of animals. To reduce the need for antibiotic use, we focus on ways to prevent bacterial disease in the first place.
Some of the key tools to disease prevention are:
- Vaccines that protect animal health; animal health companies are continuously researching new vaccines to address a variety of disease threats caused by both viruses and bacteria.
- Good nutrition that bolsters an animal’s immune system
- Biosecurity measures that protect animals from extreme weather and help stop bacteria from entering the farm
- Good animal husbandry practices that reduce animal stress, which can reduce the strength of an animal’s immune system
- Regular veterinary visits to ensure animals are closely monitored by an expert.
New tools, such as wearable sensors and A.I. powered video monitoring, are being adopted to detect early signs of disease. Some can even capture indications not visible to the human eye. This allows veterinarians to identify vulnerable animals earlier in the disease process. Using more accurate diagnostics – like big data applications and molecular diagnostics – veterinarians can identify the specific cause of the illness and target their treatment appropriately. This early intervention also allows for better isolation practices that can limit the spread of an illness to other animals.
Despite all these efforts, animals still get sick and veterinarians need to have access to the proper antibiotics to protect animal health and welfare. But we are working hard to help veterinarians and producers use a variety of tools to maintain animal health and use antibiotics as a last resort.
Science and Data are Key
Science-driven data on antibiotic use is needed to help inform and promote judicious practices
LEARN MORE
The animal health industry believes in data. It’s important for public agencies to collect science driven data on the use of antibiotics. This means there needs to be a stated scientific purpose so a data collection system can be devised to answer the scientific questions. Good data sets should also help producers and veterinarians become more careful users of antibiotics.
Good antibiotic data collection would also cover all uses of antibiotics. Currently, no single data set exists that compares antibiotic use levels across all sectors.
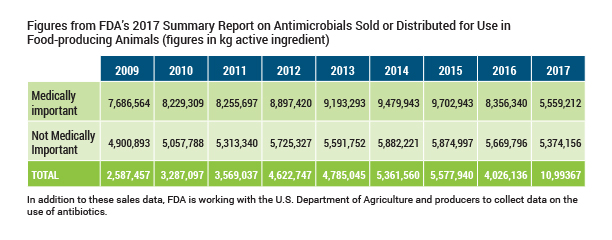
FDA, as required by law, collects information from companies on the amount of antibiotics sold each year for use in food producing animals. Sales and use are not the same, and the annual report issued by FDA lists some cautions in interpreting the sales data presented. For example, it is inappropriate to compare the volume of animal sales to human sales or use due to the number of humans in the U.S. (320 million) versus the number of food animals (approx. 10 billion) and the differences in physical characteristics of humans and animals.
The chart below shows sales results reported by FDA since this data collection began in 2009. Total sales declined 22 percent in 2017 compared to 2016 and 13 percent since 2009. Sales of medically important antibiotics declined 33 percent in 2017 compared to 2016 and 28 percent since 2009. Note that 2017 was the year that the FDA program eliminating growth promotion use of medically important compounds and mandating veterinary oversight for remaining uses went in effect.
[Insert Table Here]
The FDA and Judicious Use
Collaboration and continuous improvement contributes to strong animal health outcomes
The FDA, the animal health industry, and other stakeholders are making continuous improvement to ensure judicious use of antibiotics in animals.
LEARN MORE
In 2018 FDA announced a Five-Year Plan for Supporting Antimicrobial Stewardship in Veterinary Settings. The plan contains 32 proposed actions grouped under three broad goals:
- Align antimicrobial drug product use with the principles of antimicrobial stewardship;
- Foster antimicrobial stewardship in veterinary settings; and
- Enhance monitoring of antimicrobial resistance and antimicrobial drug use in animals.
The major components of this plan relevant to the animal health industry are:
- Revise, as necessary, the durations of use for those medically important antibiotic products with no defined duration: FDA/CVM recently announced a list of products affected by this objective and announced the availability of funding for data/information that would support FDA decisions.
- Move all remaining medically important antibiotic products from over-the-counter to prescription status.
- Revise Guidance for Industry #152, Appendix A, the list of medically important compounds.
- Develop a strategy for promoting stewardship in companion animals.
The animal health industry will continue to work with FDA to implement these actions in a way that ensures judicious use and protects animal health.